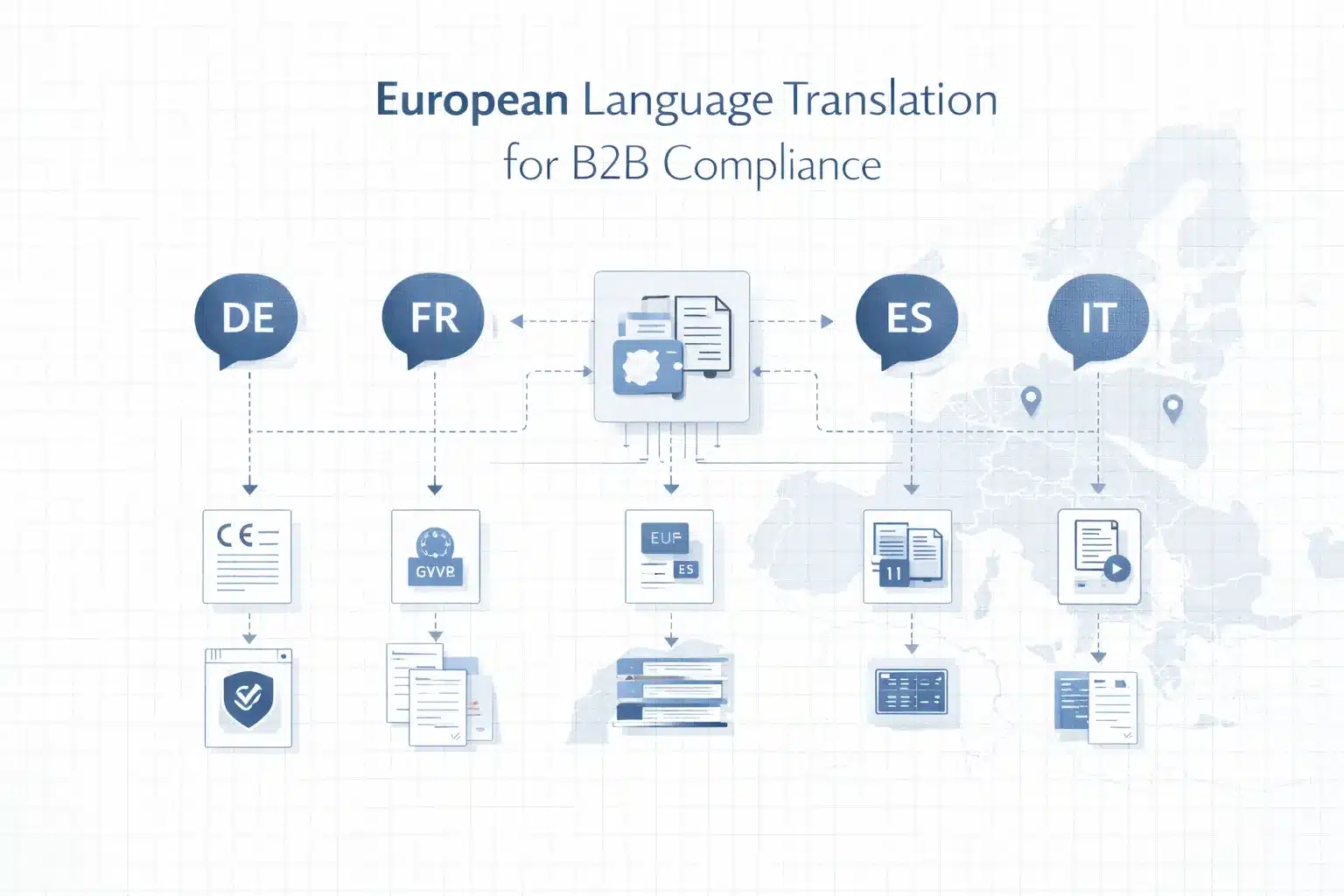
European language translation services provide professional translation across 24 EU official languages, ensuring regulatory compliance, consistency, and accuracy for B2B market entry and operations.
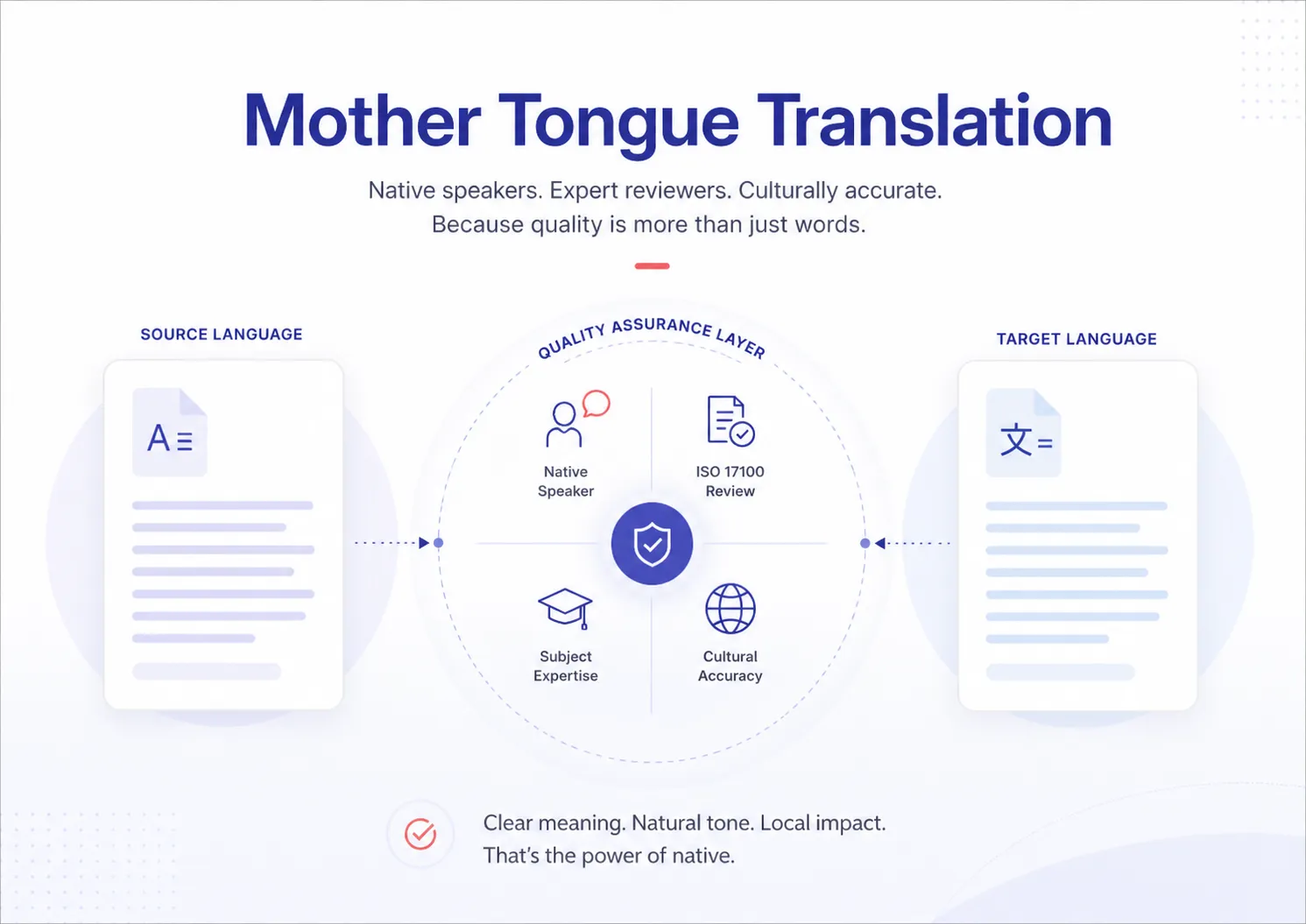
These services combine native-language expertise with structured QA processes such as ISO 17100 workflows and terminology management to deliver reliable multilingual output.
For B2B organisations, European translation is not a one-off task but a coordinated programme covering multiple languages, regulatory frameworks, and consistent terminology across legal, technical, and commercial content.
European Language Coverage: The 24 EU Official Languages and the Extended European Language Landscape
The European Union has 24 official languages, but B2B translation prioritisation depends on market size, regulatory requirements, and operational footprint rather than full language coverage.
| Language | ISO Code | Language Family | B2B Priority Tier | Primary Market | Key B2B Use Cases | Internal Link |
| German | DE | Germanic | TIER 1 | Germany, Austria, Switzerland | Contracts, compliance, technical manuals, CE marking, supply chain | /blog (German translation) |
| French | FR | Romance | TIER 1 | France, Belgium, Switzerland, Luxembourg | Legal, regulatory, EU institutional, marketing, HR | /blog (French translation) |
| Spanish | ES | Romance | TIER 1 | Spain + 20 LATAM markets | Marketing, eCommerce, legal, HR | /blog (Spanish translation) |
| Italian | IT | Romance | TIER 1 | Italy | Manufacturing, fashion, food, legal, technical | /blog (Italian translation) |
| Polish | PL | Slavic | TIER 1 | Poland | Supply chain, manufacturing, HR, legal | /blog/polish-translation |
| Dutch | NL | Germanic | TIER 2 | Netherlands, Belgium | Finance, legal, technical, eCommerce | /blog (Dutch translation) |
| Portuguese | PT | Romance | TIER 2 | Portugal + Brazil | Legal, commercial, eCommerce, HR | /blog (Portuguese translation) |
| Swedish | SV | Germanic | TIER 2 | Sweden | Technical, manufacturing, legal, regulatory | (language page) |
| Romanian | RO | Romance | TIER 2 | Romania | HR, legal, technical | (language page) |
| Czech | CS | Slavic | TIER 2 | Czech Republic | Manufacturing, automotive, technical | (language page) |
| Hungarian | HU | Finno-Ugric | TIER 3 | Hungary | Manufacturing, technical, legal | (language page) |
| Greek | EL | Hellenic | TIER 3 | Greece, Cyprus | Legal, shipping, tourism, financial | (language page) |
| Danish | DA | Germanic | TIER 3 | Denmark | Technical, maritime, pharmaceutical, food | (language page) |
| Finnish | FI | Finno-Ugric | TIER 3 | Finland | Technical, manufacturing, regulatory | (language page) |
| Bulgarian | BG | Slavic | TIER 3 | Bulgaria | Legal, HR, technical | (language page) |
| Slovak | SK | Slavic | TIER 3 | Slovakia | Automotive, manufacturing | (language page) |
| Croatian | HR | Slavic | TIER 3 | Croatia | Tourism, legal | (language page) |
| Slovenian | SL | Slavic | TIER 3 | Slovenia | Technical, automotive | (language page) |
| Lithuanian | LT | Baltic | TIER 3 | Lithuania | Legal, tech | (language page) |
| Latvian | LV | Baltic | TIER 3 | Latvia | Legal, shipping, tech | (language page) |
| Estonian | ET | Finno-Ugric | TIER 3 | Estonia | Tech, digital, legal | (language page) |
| Maltese | MT | Semitic | TIER 3 | Malta | Legal, financial services, gaming | (language page) |
| Irish | GA | Celtic | TIER 3 | Ireland | EU institutional, legal | (language page) |
The Five European Language Families and Why They Matter for Translation Planning
European language families determine structural complexity, expansion rates, and translator specialisation requirements across multilingual programmes.
Romance languages (French, Spanish, Italian, Portuguese, Romanian) share Latin roots but require separate translators despite cognates. Germanic languages (German, Dutch, Swedish) introduce compound structures, with German expanding text by 20–30%.
Slavic languages (Polish, Czech) use inflection-heavy grammar, increasing length variability. Baltic languages (Lithuanian, Latvian) require niche expertise due to smaller translator pools.
Finno-Ugric languages (Finnish, Hungarian, Estonian) create 30–50% expansion, demanding early DTP planning and specialist linguists.
Non-EU European Languages With High B2B Demand: Norwegian, Swiss, Ukrainian, Turkish, and Serbian
Non-EU European languages frequently become mandatory in B2B translation programmes due to trade, regulation, and workforce requirements.
Norwegian (Bokmål ~85% usage) is required for EEA compliance, including CE marking and food labelling. Swiss variants (CH-DE, CH-FR, CH-IT) adapt standard EU languages for Swiss markets.
Ukrainian demand has increased post-2022 for HR and compliance. Turkish supports EU trade and manufacturing supply chains. Catalan (~9 million speakers) is required for regional compliance in Spain.
Which European Languages Should a B2B Company Prioritise First? A Market-Size Decision Framework

B2B language prioritisation follows three criteria: market size, regulatory obligation, and operational footprint.
Tier 1 languages deliver maximum ROI: German (~25% EU GDP) for compliance and contracts, French for regulatory and institutional contexts, and Polish for manufacturing and workforce demand.
Tier 2 languages (Spanish, Italian, Dutch, Portuguese, Swedish, Romanian) expand coverage based on revenue and presence.
Tier 3 languages are added for full EU compliance, public procurement, or local operational requirements.
Translation Quality Assurance for European Language Programmes: Standards, Tools, and Processes
Translation quality assurance (QA) ensures translations meet defined accuracy, consistency, and compliance standards through structured processes, tools, and measurable quality thresholds.
| Standard / Tool | Type | What It Governs | Applies To | Key Requirement for EU Translation |
| ISO 17100:2015 | Process standard | Translator qualifications, revision, workflow, documentation | All professional translation | Translator + independent reviser; two-stage mandatory |
| EN 15038:2006 | Precursor standard | Same as ISO 17100 (legacy EU specs) | EU public sector | Two-stage workflow; documented competence |
| MQM | Error typology | Error classification and scoring | QA-evaluated translation | Accuracy, fluency, terminology, style categories |
| DQF | QA framework | Human + automated evaluation | Enterprise programmes | MQM + productivity metrics |
| Xbench | QA tool | Terminology, consistency, formatting | CAT workflows | Pre-delivery QA report |
| Verifika | QA tool | Tag/format checks, consistency | TMS outputs | Complementary QA validation |
| Trados QA Checker | Integrated QA | Real-time QA during translation | Trados workflows | In-segment QA + final checks |
| ISO 30042 (TBX) | Termbase standard | Terminology structure | All translation | Termbase portability across tools |
| LISA QA Model | Legacy model | Error scoring (major/minor/critical) | Enterprise legacy | Weighted error penalties |
What Translation Quality Assurance Covers: Error Typology, Severity Levels, and Pass/Fail Thresholds
Translation QA uses structured error typologies and severity scoring to define measurable quality thresholds before production begins.
MQM evaluates six categories: accuracy (mistranslation, omission), fluency (grammar, register), terminology (incorrect or inconsistent terms), style (awkward phrasing), locale conventions (dates, units), and critical errors (legal or safety risks).
Severity levels, critical, major, minor, are scored per 1,000 words. Standard B2B thresholds: 0 critical errors, ≤2 major, ≤10 minor, ensuring objective pass/fail criteria and contractual clarity.
Automated QA Tools for Translation: What Xbench, Verifika, and CAT-Integrated QA Actually Check
Automated QA tools detect rule-based errors at scale before human revision, ensuring consistency and technical accuracy.
They verify completeness (missing or untranslated segments), numerical consistency (figures, dates, units), terminology compliance against termbases, and tag integrity in structured files (HTML, XML, InDesign exports).
They also flag punctuation and spacing issues. However, they cannot assess meaning, tone, or subject accuracy, making human revision mandatory under ISO 17100 workflows.
How Medical and Pharmaceutical Translators Ensure Terminology Consistency Across Multi-Language EU Programmes
Medical translators ensure terminology consistency using termbases, translation memory, cross-language QA, and back-translation.
A validated termbase (ISO 30042 TBX) enforces INN consistency (e.g., “ibuprofen” unchanged across 24 languages) and MedDRA-approved terms. Shared TM ensures reuse of EMA QRD-approved phrases.
Cross-language QA verifies identical data across all versions. Back-translation checks high-risk sections (dosage, contraindications), ensuring zero semantic drift in regulatory submissions.
Translation Quality Assurance for EU Legal and Compliance Content: What B2B Buyers Need to Specify
EU legal translation requires explicit QA specifications to prevent compliance failures and contractual risk.
Buyers must define: (1) quality tier—ISO 17100 human translation only; no raw MT; (2) defined-term consistency via termbase; (3) back-translation for bilingual binding contracts; (4) regulatory framework alignment (GDPR, EU FIC, MDR); and (5) timeline including internal legal review.
Without these specifications, translation outputs fail regulatory, legal, or evidential standards.
B2B Use Cases for European Language Translation: Regulatory Compliance, Market Entry, and Operations
B2B European translation demand is driven by regulatory compliance, market entry expansion, and ongoing operational requirements across multiple EU languages and jurisdictions.
| Use Case Category | Typical Document Types | Languages Required | QA Standard Required | B2B Driver |
| EU regulatory compliance | CE marking documentation; EU FIC labels; GDPR notices; MDR files; SDS | All EU markets where sold | ISO 17100 + regulatory terminology | Legal obligation; market access |
| EU market entry | Website localisation; eCommerce PDPs; marketing; contracts | Tier 1 first (DE, FR, ES, IT, PL) | ISO 17100 (legal); MTPE (volume content) | Revenue growth; acquisition |
| HR and employment law | Contracts; policies; employee notices | Workforce languages | ISO 17100 (legal HR); MTPE (internal comms) | Legal compliance |
| Supply chain | Supplier agreements; POs; specs; reports | Supplier/partner languages | ISO 17100 + technical QA | Risk reduction; clarity |
| EU institutional | Tenders; bids; grants | Per tender (EU official languages) | ISO 17100; certified where required | Contract award |
| Clinical & pharma | SmPC, PIL, CTD, ICF | Up to 24 EU languages | ISO 17100 + pharma QA + back-translation | Regulatory approval |
CE Marking and EU Product Compliance Translation: Which Documents Require Which Languages
CE marking requires translation of technical documentation into the official language(s) of each EU member state where the product is marketed.
Under the Machinery Directive, Instructions for Use must be provided in local languages for each market (e.g., German, French, Italian simultaneously).
The Low Voltage Directive mandates translated safety instructions. MDR requires up to 24 language versions for medical devices. The result: manufacturers must run multi-language, parallel translation programmes with consistent terminology and DTP-ready outputs.
GDPR Translation for European Operations: Privacy Notices, Data Subject Rights, and DPA Requirements
GDPR requires that all data subject-facing content is provided in the individual’s language, not just English.
Privacy notices must meet the “clear and plain language” standard in each target language, not just literal translation. Data subject requests (DSARs) must be answered in the request language.
Employee GDPR notices must match workforce languages. DPAs may also require localisation depending on jurisdiction, making GDPR translation an ongoing compliance requirement, not a one-time task.
EU Institutional Translation Standards: What Businesses Tendering for EU Contracts Must Know
EU institutional translation requires adherence to DGT standards, ISO 17100 workflows, and official EU terminology via IATE.
The European Commission’s DGT produces ~2 million pages annually across 24 languages, setting strict benchmarks: qualified subject-matter translators, mandatory independent revision, and terminology alignment with EUR-Lex and IATE.
Businesses tendering for EU contracts must match these standards to remain compliant and competitive in procurement processes.
Commission European Language Translation With Full QA Documentation and Language Coverage Across All EU Markets
European language translation with ISO 17100 workflows, MQM-aligned QA, and full 24-language EU coverage ensures compliance, consistency, and faster market entry across regulated industries.
Whether you need German CE marking documentation, French and Italian legal translations, Polish HR materials, or a 24-language EMA submission, Circle Translations delivers accuracy with documented QA.
✓ All 24 EU languages + Norwegian, Ukrainian, Turkish, Swiss variants, Catalan
✓ ISO 17100 two-stage process (translator + reviser) with audit trail
✓ MQM QA scoring with defined thresholds and reporting
✓ Xbench/Verifika QA checks for terminology, numbers, formatting
✓ Pharma QA: MedDRA, QRD compliance, back-translation options
✓ IATE-aligned EU regulatory terminology
✓ Parallel multi-language delivery (up to 24 languages)
✓ Translation memory savings from project one (20–40% over time)
✓ Certified translation for legal and regulatory use
✓ NDA + GDPR-compliant data handling
✓ 1-hour quote turnaround
Get a European Translation Quote → View Translation and Localisation Services→
European Language Translation Services — Frequently Asked Questions
How many official languages does the EU have and do I need all of them?
The EU has 24 official languages, but most B2B companies only need a subset based on regulatory scope and market priorities. Full coverage applies to EMA submissions, CE marking across all EU markets, and EU tenders, while most commercial expansions start with German, French, Spanish, Italian, and Polish, then scale based on revenue, workforce, and compliance obligations.
What is translation quality assurance (QA) and what does it include?
Translation QA ensures accuracy, consistency, and fitness-for-purpose through automated checks and human revision. It includes QA tools like Xbench and Verifika (terminology, numbers, tags), ISO 17100 independent revision, termbase validation, and QA reporting, with additional domain-specific checks such as MedDRA compliance and back-translation for regulated content.
What is the difference between ISO 17100 and the older EN 15038 translation standard?
ISO 17100 is the current global standard that replaced EN 15038 while maintaining equivalent core requirements. Both require qualified translators, independent revision, and documented workflows, but ISO 17100 applies internationally, and compliance with it satisfies legacy EN 15038 requirements still referenced in older EU procurement contracts.
What is MQM and how is it used in translation quality assessment?
MQM is a structured framework that categorises and scores translation errors by type and severity. It evaluates Accuracy, Fluency, Terminology, Style, and Locale errors using Critical, Major, and Minor levels, enabling objective measurement of translation quality as errors per 1,000 words against agreed thresholds.
Does Circle Translations cover all 24 EU official languages?
Yes, Circle Translations provides professional translation across all 24 EU official languages using native subject-matter experts. Multi-language programmes use centralised project management, shared termbases, and parallel workflows with cross-language QA checks to ensure consistency across all language versions.
What is IATE and how does it affect EU regulatory translation quality?
IATE is the EU’s official terminology database and the authoritative source for regulatory translation vocabulary. It ensures that translations align with EU-approved legal and regulatory terms across all languages, which is essential for GDPR, CE marking, and pharmaceutical submissions.
Can translation into European languages be done with machine translation (MT)?
Machine translation is suitable only for high-volume, low-risk content and must be post-edited for professional use. MTPE works for eCommerce and internal content but is not appropriate for legal, regulatory, HR, or safety-critical materials, where full human translation and “no raw MT” must be specified.
How long does a multi-language EU translation programme take?
EU translation timelines typically range from 5 to 25 business days depending on volume, languages, and QA requirements. For example, 5-language CE marking takes 7–15 days, 24-language EMA submissions take 15–25 days, and GDPR notices into 10 languages take 5–8 days, with parallel workflows reducing delivery time.
What is the difference between certified and sworn translation for EU regulatory submissions?
Certified translation confirms accuracy via a signed statement, while sworn translation has legal authority from a government-recognised translator. Certified translation is sufficient for most EU regulatory use, while sworn translation is required for specific national legal processes such as court submissions in countries like Germany, France, and Poland.