Food label translation services deliver regulatory-compliant translation of packaging copy, ingredient lists, allergen declarations, nutrition panels, and health claims, ensuring food products meet EU FIC (Regulation 1169/2011), FDA 21 CFR Part 101, and global labelling laws across export markets.
Food labelling is a legal requirement, not a marketing task. Every export market mandates specific elements, formats, and languages, including ingredients, allergens, nutrition data, and origin declarations. Non-compliance results in customs delays, product recalls, fines, and reputational damage.
Circle Translations delivers food label translation as a compliance system—combining food regulatory expertise, CAT tools, QA workflows, and multilingual DTP. This ensures accurate allergen handling, correct nutrition claim wording, and fully compliant packaging across EU, US, UK, and global markets.
Mandatory Food Label Requirements by Market: EU, US, UK, and Key Export Destinations
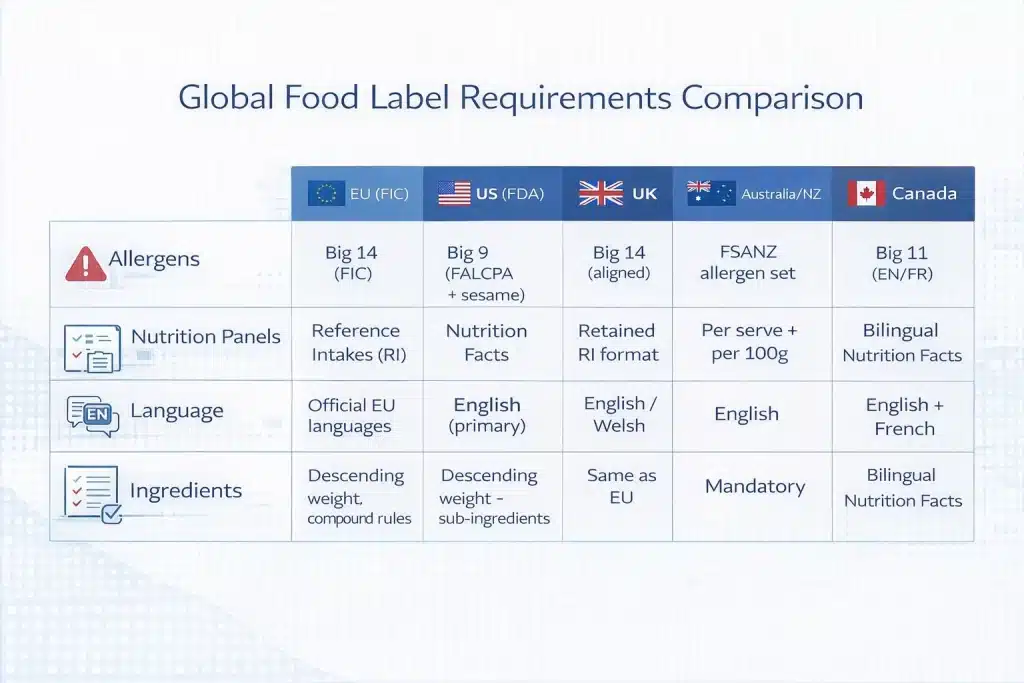
Food label requirements differ by market, with each jurisdiction defining mandatory elements, formats, and language rules that directly affect translation and packaging compliance.
Mandatory Elements by Market
| Mandatory Element | EU (FIC — Reg. 1169/2011) | US (FDA 21 CFR Part 101 + USDA) | UK (Food Information Regulations 2014) | Australia/NZ (FSANZ) | Canada (FDR / SFCR) |
| Name of the food | Legal/customary name required | Statement of identity required | Same as EU (with divergence) | Mandatory | Mandatory |
| Ingredients list | Descending weight; compound rules | Descending weight; sub-ingredients | Same as EU | Mandatory | Mandatory (EN/FR) |
| Allergen declaration | Big 14 highlighted | Big 9 (FALCPA + sesame) | Big 14 (aligned) | FSANZ allergen set | Big 11 (EN/FR) |
| Nutrition info | Reference Intakes (RI) | Nutrition Facts panel | RI format retained | Per serve + per 100g | Bilingual Nutrition Facts |
| Net quantity | Metric units | Metric + US customary | Metric | Metric | Metric (+ some imperial) |
| Date marking | “Best before” / “Use by” | Flexible formats | Same as EU | Mandatory | Mandatory |
| Country of origin | Mandatory for some categories | Mandatory for specific foods | UK-specific rules | Mandatory | Mandatory |
| Language | Official EU languages | English (primary) | English / Welsh | English | Mandatory EN/FR |
EU Food Information to Consumers Regulation (FIC 1169/2011): What Every Exporter Must Know
EU FIC 1169/2011 defines mandatory language, format, and content requirements for all food sold in EU markets. Labels must appear in the official language of each target country.
Minimum font size is 1.2mm, forcing DTP adjustments for languages with 20–30% expansion. QUID percentages must be correctly placed, and compound ingredient rules must be applied.
Regulated phrases like “best before” must use approved translations. Non-compliance leads to market rejection.
FDA Food Label Requirements Under 21 CFR Part 101: Nutrition Facts, Serving Size, and Bilingual Labelling
FDA 21 CFR Part 101 mandates a fixed Nutrition Facts format with specific nutrients, serving size rules, and Daily Value calculations. EU Reference Intakes cannot be reused.
US labels must include cholesterol and trans fat, even if absent in EU labels. Bilingual labels must include full English equivalents. Incorrect nutrient formats or terminology create immediate compliance failure.
UK Post-Brexit Food Labelling: What Has Changed from EU FIC and What Remains Aligned
UK food labelling follows retained EU law with key divergences in authority and allergen rules. Great Britain uses UK Food Information Regulations with FSA oversight, while Northern Ireland follows EU FIC.
Natasha’s Law requires full allergen labelling for PPDS foods. “Product of UK” claims are now UK-specific. Dual compliance is required for EU and UK distribution.
Canada and Australia/NZ Food Labelling: Bilingual Requirements, FSANZ Standards, and Import Compliance
Canada mandates full bilingual English/French labels, while Australia/NZ follow FSANZ standards with distinct nutrition and allergen rules.
Canada requires bilingual Nutrition Facts tables and all mandatory elements in both languages. FSANZ requires per serve and per 100g nutrition panels and a defined “Contains” allergen format.
These structural differences require separate label versions, not direct translation reuse.
Allergen Declarations and Nutrition Claims in Translated Food Labels: Market-Specific Rules and Translation Accuracy Requirements
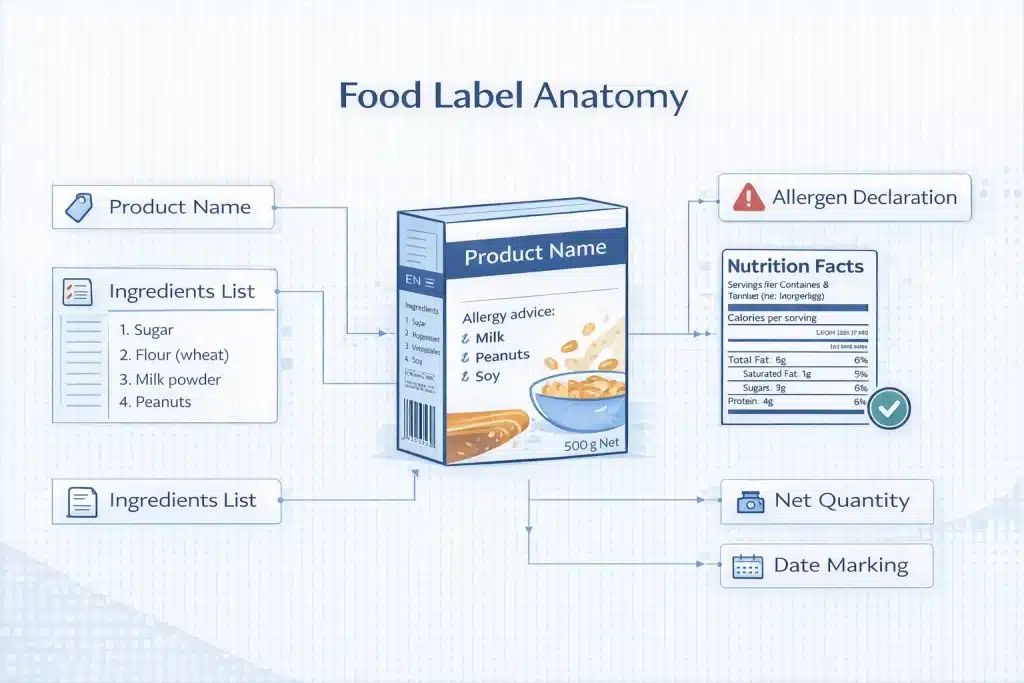
Allergen declarations and nutrition or health claims carry the highest compliance risk in food label translation, because errors directly impact consumer safety and regulatory approval.
Allergen Declaration Requirements by Market
| Allergen | EU Big 14 (FIC 1169/2011) | US Big 9 (FALCPA + FASTER Act 2023) | UK (aligned with EU Big 14) | Australia/NZ (FSANZ) | Canada (Big 11) |
| Cereals containing gluten | ✅ Mandatory | Wheat only ✅ | ✅ Mandatory | ✅ (gluten) | ✅ Wheat + gluten grains |
| Crustacea | ✅ Mandatory | ✅ (Shellfish incl. molluscs) | ✅ Mandatory | ✅ Separate | ✅ Mandatory |
| Eggs | ✅ Mandatory | ✅ Mandatory | ✅ Mandatory | ✅ Mandatory | ✅ Mandatory |
| Fish | ✅ Mandatory | ✅ Mandatory | ✅ Mandatory | ✅ Mandatory | ✅ Mandatory |
| Peanuts | ✅ Mandatory | ✅ Mandatory | ✅ Mandatory | ✅ Mandatory | ✅ Mandatory |
| Soybeans | ✅ Mandatory | ✅ Mandatory | ✅ Mandatory | ✅ Mandatory | ✅ Mandatory |
| Milk | ✅ Mandatory | ✅ Mandatory | ✅ Mandatory | ✅ Mandatory | ✅ Mandatory |
| Tree nuts | ✅ Each named | ✅ Category | ✅ Mandatory | ✅ Mandatory | ✅ Mandatory |
| Celery | ✅ Mandatory | ❌ Not required | ✅ Mandatory | ❌ Not required | ❌ Not required |
| Mustard | ✅ Mandatory | ❌ Not required | ✅ Mandatory | ❌ Not required | ✅ (seeds) |
| Sesame | ✅ Mandatory | ✅ (since 2023) | ✅ Mandatory | ✅ Mandatory | ✅ Mandatory |
| Sulphites (>10 mg/kg) | ✅ Mandatory | ❌ Not required | ✅ Mandatory | ✅ Mandatory | ✅ Mandatory |
| Lupin | ✅ Mandatory | ❌ Not required | ✅ Mandatory | ✅ Mandatory | ❌ Not required |
| Molluscs | ✅ Mandatory | Included in shellfish | ✅ Mandatory | ✅ Mandatory | ✅ Mandatory |
How EU FIC Allergen Highlighting Works and Why Translation Must Preserve the Highlighting Format
EU FIC requires allergens to be visually distinguished within the ingredients list, and translation must preserve this formatting exactly. Highlighting (bold, italic, underline) must appear in every instance, including compound ingredients.
File formats must retain styling, requiring InDesign, Illustrator, or structured formats. Translators must identify allergen triggers, not just translate text. Incorrect highlighting or loss of formatting creates compliance failure and consumer safety risk.
As European Commission (2014) explains in Guidance on the Provision of Food Information to Consumers (Regulation (EU) No 1169/2011), “Allergens must be emphasized through a typeset that clearly distinguishes them from the rest of the list of ingredients,” highlighting that both content and formatting are mandatory for compliance and must be preserved across all representations, including translations.
Nutrition Claims and Health Claims on Food Labels: EU Regulation 1924/2006, FDA Authorised Claims, and Translation Rules
Nutrition and health claims must use authorised regulatory wording, not free translation, in every market. EU Regulation 1924/2006 and EFSA define fixed claim language. FDA 21 CFR 101 governs US claims with strict nutrient thresholds.
Claims like “low fat” or “high protein” must meet numeric criteria and approved wording. Health claims require exact authorised phrasing. Translating claims incorrectly creates regulatory violations even when the underlying data is accurate.
As the European Commission (2012) explains in its implementation guidance for Regulation (EC) No 1924/2006, “Only nutrition and health claims that comply with the conditions laid down in the Regulation and are included in the lists of authorised claims may be used,” highlighting that both wording and usage are strictly controlled and cannot be freely translated.
Organic, Free-From, and Sustainability Claims: Market-Specific Certification Requirements and Translation Accuracy
Certification claims such as “organic” and “gluten-free” require regulated terminology that differs by language and market. EU organic terms must use approved equivalents like “bio” or “ökologisch,” not literal translations.
The FDA and the USDA regulate US organic claims. “Gluten-free” is capped at 20 ppm in both the EU and the US but requires exact wording. Misuse of certification terminology creates legal exposure and invalid claims.
Multilingual Food Label DTP and Packaging Artwork: Text Expansion, Font Size Compliance, and Multi-Market Label Packs
Multilingual food label compliance depends on correct DTP implementation, where translated text must fit packaging constraints while maintaining regulatory font size, formatting, and layout rules.
Text Expansion by Language and DTP Impact
| Language | Approximate Text Expansion vs English | DTP Impact for Food Labels | Key DTP Risk |
| German | +20–30% | Ingredient lists expand; compound words increase length | Long compound nouns break layout |
| French | +15–20% | Bilingual EN/FR panels increase space usage | Regulated phrases significantly longer |
| Spanish | +15–20% | Moderate expansion | Regional variation affects length |
| Italian | +10–15% | Moderate expansion | Longer grammatical structures |
| Polish | +20–30% | Significant expansion | Complex inflections increase length |
| Finnish | +30–50% | Very high expansion | Extremely long compound words |
| Dutch | +15–25% | Moderate-high expansion | Compound word overflow |
| Japanese | -20–30% | Space savings | Font rendering and encoding issues |
| Chinese (Simplified) | -15–25% | Compact layout | GB standards and encoding constraints |
| Arabic | Variable (RTL) | Full layout redesign required | RTL cannot reuse LTR templates |
How to Structure a Multi-Language Food Label Without Violating Minimum Font Size Requirements
EU FIC requires a minimum 1.2mm x-height, forcing layout redesign when multiple languages increase text volume. Multi-language labels often contain 3–4× more content than English-only versions.
Solutions include larger packaging, mandatory-only multilingual panels, or compliant overstickers.
Fonts must support Latin Extended, Cyrillic, and Greek sets where required. Layout strategies prioritise ingredient lists, use language identifiers, and apply symbols to reduce text load while maintaining compliance.
Food Label Stickers and Overstickers for Market-Specific Compliance: When and How to Use Them
Overstickers provide a compliant, cost-controlled solution for adding market-specific language without reprinting packaging.
They are used for market entry, low-volume exports, and recipe updates. EU FIC requires full mandatory content, 1.2mm font size, and no obstruction of base labels.
Circle Translations applies a controlled workflow: translation, allergen validation, claims review, and DTP production, delivering print-ready files aligned with target-market regulations.
Ingredient List Translation: INCI Names, E-Numbers, and the Compound Ingredient Expansion Rule
Ingredient list translation requires strict handling of additives, compound ingredients, and naming conventions under EU and international regulations.
E-numbers remain unchanged, while additive names must be translated per language. Compound ingredients above 2% require full sub-ingredient expansion.
Supplement products may require INCI, IUPAC, or common names depending on the market. Incorrect handling creates compliance errors and mislabelled products.
Commission Food Label Translation That Passes Regulatory Review in Every Export Market
One incorrect allergen, one non-compliant claim, or one formatting error can stop your product at customs or trigger a recall. Circle Translations eliminates that risk with a controlled, audit-ready translation and DTP system.
✓ Regulatory coverage across all target markets — EU FIC (1169/2011), FDA 21 CFR Part 101, UK FSA rules, CFIA Canada bilingual labelling, FSANZ Australia/NZ, plus GCC and India frameworks
✓ Allergen control system — every ingredient cross-checked against EU Big 14, FDA Big 9, FSANZ, and CFIA Big 11 before approval
✓ Claims validation layer — EFSA-approved health claims, FDA-authorised wording, and regulated “organic” and “gluten-free” terminology enforced
✓ DTP compliance built-in — 1.2mm font size verified, allergen highlighting preserved, and print-ready artwork delivered in InDesign, Illustrator, and PDF
✓ Multi-language scalability — 2–24+ languages delivered in one workflow with a unified termbase and QA process
✓ Text expansion engineered — German (+25%), Polish (+25%), Finnish (+40%) managed without layout failure; Chinese and Japanese optimised for space
✓ Oversticker solutions — rapid market entry with compliant, print-ready multilingual labels
✓ Ingredient-level accuracy — E-numbers, compound ingredients, and INCI naming controlled per regulation
✓ Cost efficiency — translation memory delivers 20–40% savings across product portfolios within 6 months
✓ Full confidentiality — NDA protection for formulations, recipes, and packaging data
Tell us your product, target markets, and packaging format, and Circle Translations will design a compliant label translation system within 1 business hour.
Get a Food Label Translation Quote →
View Technical Translation Services →
View Translation and Localisation Services →
Food Label Translation Services — Frequently Asked Questions
What are the mandatory elements required on a food label in the EU?
EU FIC (Regulation 1169/2011) requires 11 mandatory elements on food labels. These include product name, ingredients list, allergen declaration (Big 14), net quantity, date marking, storage instructions, business operator details, origin (where required), usage instructions, alcohol content (>1.2%), and nutrition declaration with energy and six key nutrients.
How much does food label translation cost?
Food label translation costs £80–£250 per language, with DTP adding £150–£400 per language. Costs depend on word count (300–800 words), number of languages, and compliance checks. Multi-language label packs reduce costs by 20–40% through translation memory and volume pricing.
Can I use one multilingual label for all EU markets, or do I need separate labels per country?
A single multilingual label is compliant if all mandatory elements appear in each required EU language. It must meet the 1.2mm font size rule. Markets with multiple official languages require both (e.g., Belgium, Finland). Many brands combine core markets with dedicated labels and use multi-language variants for smaller regions.
What allergens must be listed on a food label in the UK?
UK law requires the declaration of 14 allergens, including gluten cereals, milk, eggs, nuts, soy, fish, and sulphites. These must be highlighted in the ingredients list. Natasha’s Law (2021) extends this to PPDS foods, requiring full ingredient and allergen labelling for products prepared and packaged on-site.
Does food label translation need to be certified?
Certified translation is not generally required for EU or US labels, but applies in specific regulatory or import contexts. Some markets require certification for product registration or customs clearance. ISO 17100-compliant translation with full QA documentation meets standard commercial requirements.
What is QUID, and why does it affect food label translation?
QUID requires a percentage declaration of key ingredients referenced in the product name or imagery. The percentage remains unchanged in translation, but placement must be correct in every language. Incorrect positioning or omission creates compliance failure under EU FIC.
How does food label translation differ for e-commerce food brands selling on Amazon or Shopify internationally?
E-commerce food brands must comply with both physical label laws and platform-specific requirements. Amazon EU requires allergen information in local languages. Canada requires EN/FR bilingual listings. Product pages and packaging must align, or listings can be removed and products blocked.
What is the difference between a ‘best before’ date and a ‘use by’ date in food labelling translation?
“Best before” refers to quality, while “use by” defines safety and legal consumption limits. Each has fixed translations (e.g., German: “Mindestens haltbar bis” vs. “Zu verbrauchen bis”). Misuse creates food safety risk and regulatory non-compliance.
Can Circle Translations handle food label DTP alongside translation?
Circle Translations provides integrated translation and DTP for food labels in a single workflow. Text is translated, formatted in InDesign or Illustrator, and checked for font size, allergen highlighting, and layout expansion. Deliverables include print-ready PDFs and native artwork files for production.