A legal and medical terms translator does not rely on dictionaries. Professional translation depends on a managed termbase that enforces approved terminology across contracts, clinical documents, and regulatory submissions, ensuring consistency, compliance, and audit-ready accuracy in legal, pharmaceutical, and healthcare contexts.
When B2B buyers search for a “legal medical terms translator,” they are evaluating three things: a specialist translator, a terminology tool, or a controlled quality system.
Terminology management is the decisive factor, because inconsistent vocabulary creates legal ambiguity in contracts and regulatory risk in clinical and pharmaceutical documentation.
In legal translation, inconsistent definitions of terms change the contractual meaning and create enforceability risks. In life sciences translation, incorrect MedDRA terms, INN misuse, or outdated terminology trigger regulatory rejection and patient safety exposure.
Circle Translations implements terminology as a system, termbases (TBX), CAT tool enforcement, QA workflows, and corpus-based extraction, to deliver consistent, compliant, and inspection-ready translation across all documents and languages.
Why Terminology Inconsistency Is the Most Costly Translation Error in Legal and Medical Contexts

Terminology inconsistency creates contractual ambiguity, regulatory rejection, and patient safety risk in legal and medical translation, where controlled vocabulary determines enforceability and compliance.
Terminology Failure Types and Consequences
| Terminology Error Type | Legal Consequence | Medical / Life Sciences Consequence | Prevention Mechanism |
| Same concept, different terms across sections | Contractual ambiguity—courts interpret each term independently; “shall,” “must,” and “is required to” create different obligations | Inconsistent MedDRA coding across adverse event reports; same event mapped to different LLTs affects signal detection | Approved preferred terms enforced via CAT tools; forbidden variants blocked in QA |
| Wrong term (synonym for a defined term) | Defined term loses its contractual meaning; synonym creates interpretation gap | INN replaced with chemical or brand name; regulatory classification mismatch | INN list integrated into termbase; automatic substitution prevention |
| False cognate / false friend | Misleading legal equivalence across jurisdictions; incorrect liability scope | Patient-facing errors; e.g., “intoxication” misinterpreted as alcohol instead of poisoning | False friends list per language pair; enforced in QA tools |
| Context-dependent polysemy | “Party” misinterpreted without legal context; incorrect entity definition | “Depression” coded to wrong MedDRA concept; clinical vs non-clinical confusion | Context-tagged termbase; domain-specific entries with disambiguation |
| Missing standardisation across multi-language dossier | Cross-border enforceability disputes; inconsistent defined terms across language versions | EMA queries due to inconsistent adverse event terminology in MAA dossiers | Centralised multi-language termbase; cross-language QA checks |
| Outdated term usage | Superseded legal terminology post-regulatory change (e.g., Brexit updates) | Deprecated MedDRA terms used; version mismatch triggers regulatory issues | Version-controlled termbase; MedDRA updates (March/September) enforced |
How Defined Terms in Legal Contracts Create Unique Translation Accuracy Requirements
Defined terms require absolute consistency because they override natural language variation and carry fixed contractual meaning. Contracts assign specific meanings to terms like “Confidential Information,” “Effective Date,” and “Material Adverse Effect.”
Any variation creates new legal concepts. Circle Translations extracts defined terms at project start, enforces approved equivalents via termbases and CAT tools, and validates every instance during revision.
Multi-language contracts include cross-language consistency checks to ensure identical legal meaning across all versions.
Medical Terminology Systems: MedDRA, ICD-11, SNOMED CT, and Why Each Has Different Translation Rules
Medical translation requires strict adherence to regulated terminology systems, each with defined usage rules and update cycles. MedDRA governs pharmacovigilance and requires approved PT and LLT usage with biannual updates.
ICD-11 supports clinical diagnosis coding using WHO-approved terms. SNOMED CT enables EHR interoperability with national-language constraints.
WHO ATC classifies drugs using INN, which is not translated. Using non-approved terms creates coding errors, regulatory rejection, and data inconsistency.
Legal System Concepts That Do Not Have Direct Equivalents: How Functional Equivalence Handles Non-Translatable Terms
Functional equivalence ensures legal accuracy when direct term translation is impossible across legal systems. Concepts like “trust,” “consideration,” and “equity” lack civil law equivalents. Translators must select jurisdiction-specific equivalents or retain the source term with an explanation.
Termbases store these decisions with legal system tags, usage notes, and forbidden alternatives. This prevents legal misinterpretation and ensures enforceability across jurisdictions.
How a Professional Terminology Management System Works: From Term Extraction to Enforced Consistency
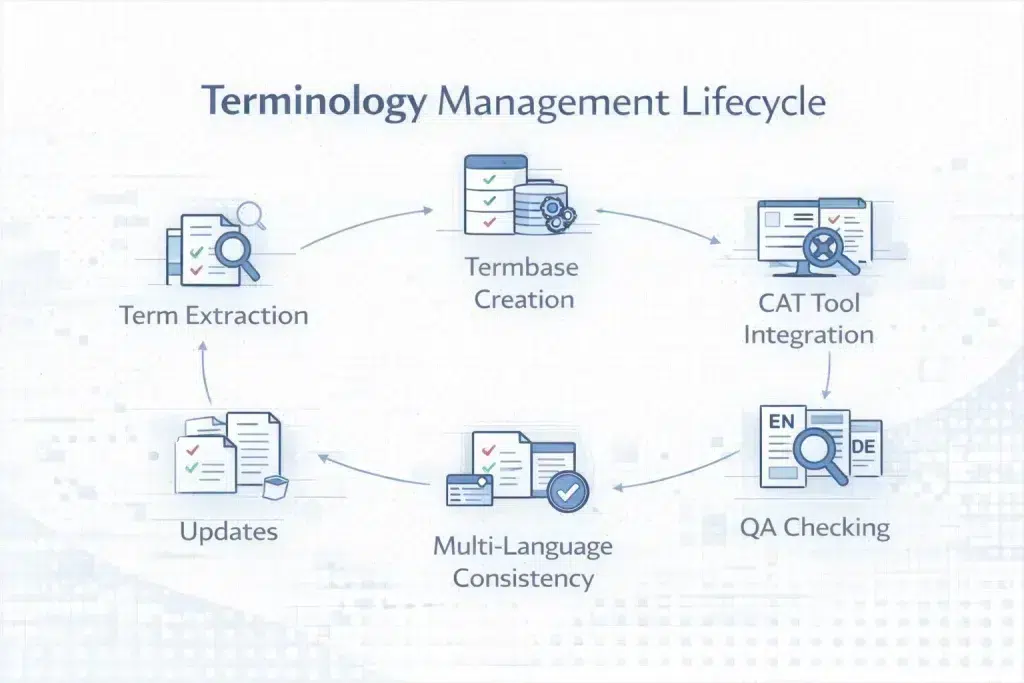
Terminology management is a structured quality system with defined inputs, controlled workflows, and enforced outputs, not a static glossary or spreadsheet.
Terminology Management Lifecycle
| Stage | What Happens | Tool / Process | Output | B2B Value |
| 1. Term extraction | Source documents scanned for high-frequency terms, defined terms, and regulatory vocabulary | SDL MultiTerm Extract, memoQ, Trados; corpus tools + manual review | Candidate term list | Captures full terminology scope; identifies hidden patterns |
| 2. Term validation | Legal, medical, or regulatory experts approve or reject terms using authoritative references | SME validation; MedDRA browser; EMA QRD; Black’s Law Dictionary | Approved term list | Ensures regulatory correctness; removes ambiguity |
| 3. Termbase construction | Terms structured with fields: source, target, domain, status, definition, context | MultiTerm, memoQ, XTM; TBX (ISO 30042) format | Structured termbase | Machine-readable; reusable across systems |
| 4. CAT tool integration | Termbase embedded into translation environment with real-time suggestions and error flags | Trados, memoQ, Phrase, XTM Cloud | Enforced terminology during translation | Prevents inconsistency at source |
| 5. QA term checking | Automated QA scans for term usage, forbidden terms, INN/MedDRA accuracy | Xbench, Verifika, CAT QA modules | QA deviation report | Detects systematic errors across large files |
| 6. Termbase update | New terms added; approved terms refined; version control applied | PM + client review workflows | Updated termbase | Continuous improvement; higher TM leverage |
| 7. Multi-language cross-check | Cross-language validation ensures consistent rendering across all target languages | Automated comparison tools | Consistency report | Eliminates multi-vendor inconsistency risk |
Building a Legal Termbase: Defined Terms, Jurisdiction Tags, and Forbidden Equivalents
A legal termbase encodes legal meaning, jurisdiction, and usage constraints, not just translations. Each entry includes source term, grammatical data, domain, and legal system tags such as common law or civil law.
Target entries define preferred equivalents, jurisdiction notes, and forbidden terms enforced in QA. Regulatory references such as GDPR Article 4(7), anchor definitions.
Circle Translations adds client-specific preferences, negotiated bilingual terms, and jurisdiction updates to maintain enforceability across evolving legal frameworks.
Life Sciences Termbase: MedDRA Updates, INN Lists, and Multi-Language Consistency Across a Regulatory Dossier
A life sciences termbase integrates multiple regulated systems and enforces version-controlled terminology across large-scale dossiers. INN fields apply “do not translate” rules for active substances.
MedDRA terms are updated twice annually and include PT, LLT, and SOC structures. EMA QRD templates standardise labelling language. WHO-UMC causality terms and ICH E2A safety criteria enforce exact phrasing.
Deprecated and forbidden terms are blocked in QA. This structure ensures consistency across 100,000+ word CTDs and multi-language submissions.
How Corpus-Based Term Extraction Builds a Termbase From Your Existing Document Archive
Corpus-based term extraction converts existing translated documents into a validated, production-ready termbase. A parallel corpus aligns source and target texts to identify term frequency, variation, and inconsistencies.
Extraction tools generate candidate lists, which subject matter experts validate. Approved terms are structured in TBX format and deployed via CAT tools.
Clients with 5–10 years of contracts or clinical reports achieve immediate gains: 30–60% TM leverage and measurable terminology consistency across all future projects.
Terminology QA in Legal and Life Sciences Translation: Automated Checking, Human Review, and Forbidden Terms
Terminology accuracy requires real-time enforcement, automated QA, and independent human review, because no single layer detects all legal, regulatory, and semantic risks.
QA System Layers
| QA Layer | What It Checks | Tool | When Applied | Catches |
| Termbase enforcement (CAT integration) | Real-time validation of preferred terms; forbidden terms flagged as errors | Trados, memoQ, XTM with integrated termbase | During translation | Terminology errors at source before entering draft |
| Automated QA (Xbench/Verifika) | Term consistency, numbers, punctuation, tags, and INN presence | Xbench, Verifika, Trados QA Checker | After translation, before revision | Systematic errors across large files (e.g., 50,000+ words) |
| Independent revision (ISO 17100) | Full bilingual review for meaning, terminology, fluency, and formatting | Qualified second linguist | After QA | Contextual and legal-system errors |
| Legal/medical subject matter review | Domain validation of clinical, regulatory, or legal accuracy | External SME (lawyer, pharmacist, regulator) | High-risk documents only | Domain-specific inaccuracies beyond translation |
| Back-translation | Reverse translation to detect meaning shifts | Independent back-translator | COA/PRO, high-risk labelling | Hidden semantic deviations (“unknown unknowns”) |
How Forbidden Terms Lists Prevent the Most Damaging Terminology Errors in Legal and Medical Translation
Forbidden terms eliminate high-risk errors by blocking plausible but incorrect terminology before delivery. These terms are linguistically valid but legally or regulatorily wrong. In pharmaceutical translation, forbidden terms include non-approved MedDRA synonyms, outdated PTs, and incorrect INN substitutions.
In legal translation, they include synonyms for defined terms and jurisdictionally incorrect equivalents. CAT tools flag forbidden terms as critical errors, and no project passes QA without documented resolution or justified exception.
Life Sciences Translation Quality: How ISO 17100, ISO 30042, and ICH Guidelines Interact in a Single Programme
Life sciences translation quality depends on simultaneous compliance with ISO 17100, ISO 30042, and ICH guidelines. ISO 17100 enforces translator competence, two-stage revision, and audit trails.
ISO 30042 defines TBX termbase structure for interoperability and inspection readiness. ICH E6, E3, Q10, and M4 define document content, regulatory expectations, and risk-based quality levels.
Circle Translations aligns all three: structured workflows, TBX termbases, and guideline-specific translator assignment ensure regulatory compliance.
How AI-Assisted Terminology Management Improves Speed Without Compromising Regulatory Accuracy
AI accelerates terminology extraction and consistency detection, but human validation ensures regulatory correctness and auditability. AI identifies term candidates, detects inconsistencies, and suggests context-aware terminology in CAT tools.
However, AI cannot validate MedDRA compliance, determine legal functional equivalence, or produce GMP-compliant audit trails. Circle Translations uses AI for speed and scale, while qualified legal and medical experts approve all terminology decisions and maintain full validation records.
Build the Terminology Management System Your Legal and Life Sciences Translation Programme Needs
Translation without a controlled terminology system creates legal ambiguity, regulatory rejection, and audit failure. Circle Translations implements structured terminology systems that eliminate these risks.
✓ Corpus-based term extraction — convert existing contracts, CTDs, and reports into validated termbases
✓ Legal term bases — defined terms, jurisdiction tags, forbidden equivalents enforced across all documents
✓ Life sciences termbases — MedDRA (biannual updates), INN control, EMA QRD templates, WHO-UMC terms
✓ ISO 30042 TBX format — portable, machine-readable termbases integrated with all major TMS platforms
✓ CAT tool enforcement — real-time preferred term suggestions and hard-stop forbidden term validation
✓ Automated QA (Xbench, Verifika) — full terminology consistency scan before human revision
✓ ISO 17100 workflow — mandatory independent revision with documented audit trail
✓ AI-assisted extraction — accelerated processing with expert-validated terminology decisions
✓ Multi-language consistency checks — identical concept rendering across all target languages
✓ Version-controlled termbase — full history with validator, timestamp, and regulatory reference
Tell us your domain, document types, and target languages. Circle Translations will design a terminology management system aligned with your compliance and operational requirements.
Request a Terminology Programme Consultation →
View Corpus Translation Services →
View Technical Translation Services →
Legal and Medical Terms Translation — Frequently Asked Questions
What is the difference between legal translation and general translation?
Legal translation requires strict consistency of defined terms, legal system knowledge, and enforceable accuracy. Unlike general translation, it operates within contracts and statutes where wording defines obligations. Errors create legal ambiguity, unenforceability, or liability, not just readability issues.
Why is legal and medical translation more difficult than other types of translation?
Legal and medical translation is harder because both use controlled vocabularies with non-interchangeable terms. Synonyms create errors. Legal systems differ across jurisdictions, and medical content must follow standards like MedDRA or ICD-11. Accuracy depends on domain rules, not language fluency.
What qualifications does a professional legal or medical translator need?
Professional translators require domain degrees or certifications plus ISO 17100-compliant competence. Legal translators hold law-related qualifications or certifications (e.g., CIOL, ATA). Medical translators hold life sciences degrees. Procurement should require documented subject-matter expertise, not just language skills.
What is a termbase, and how is it different from a translation memory?
A termbase stores approved terminology, while a translation memory (TM) stores full translated segments. Termbases enforce vocabulary consistency. TMs improve efficiency through reuse. Combined, they reduce costs by 30–70% and maintain accuracy across large-scale translation programmes.
Can machine translation handle legal and medical terminology accurately?
Raw machine translation cannot reliably handle regulated terminology. It lacks controlled vocabulary enforcement and fails in context. MTPE works for low-risk content when combined with termbases and human review. Do not use MT alone for contracts, regulatory submissions, or medical labelling.
What is life sciences translation, and how does it differ from medical translation?
Life sciences translation covers regulated pharmaceutical and clinical content, while medical translation is broader. Life sciences include CTDs, trials, and labelling under EMA, FDA, and ICH rules. Medical translation includes general healthcare materials without the same regulatory constraints.
How often should a legal or life sciences termbase be updated?
Life sciences term bases are updated at least twice yearly with MedDRA releases. They also update after each project. Legal term bases update when laws change, jurisdictions shift, or new document types are added. Continuous updates improve consistency and reduce error rates.
What is corpus translation, and how does it help build terminology databases?
Corpus translation uses existing bilingual documents to extract and validate terminology. Parallel texts identify term usage, frequency, and inconsistencies. This builds a reusable termbase from historical data, reducing costs and improving consistency across future translation projects.
How does Circle Translations manage terminology for clients with multi-language legal programmes?
Circle Translations uses a centralised multi-language termbase with cross-language QA checks. Each source term has approved equivalents across all languages. Consistency reports ensure identical meaning across versions, preventing legal discrepancies and ensuring enforceability in multi-jurisdiction documents.