Translating medical documents online is possible, but it carries significant risk if the wrong tool or vendor is used. Free AI tools such as Google Translate and ChatGPT are not HIPAA or GDPR compliant and may store or process submitted patient data. For personal reference, they may be acceptable, but for clinical, regulatory, or B2B use, professional medical translation is required.
Healthcare organisations, clinics, insurers, and clinical trial sponsors face the same operational challenge: balancing translation speed with patient data protection, medical accuracy, and regulatory compliance.
Uploading medical records to a free translation tool may appear convenient, but it can expose Protected Health Information (PHI) and create clinical risk through translation errors.
This guide explains which medical documents can be translated online safely, when AI tools fail, and what a compliant medical translation workflow looks like.
It also outlines the specific privacy risks under HIPAA and GDPR, compares translation methods such as machine translation post editing (MTPE) and human medical translation, and provides a decision framework for choosing the correct approach for different medical document types.
Types of Medical Documents That Require Professional Translation and the Stakes for Each
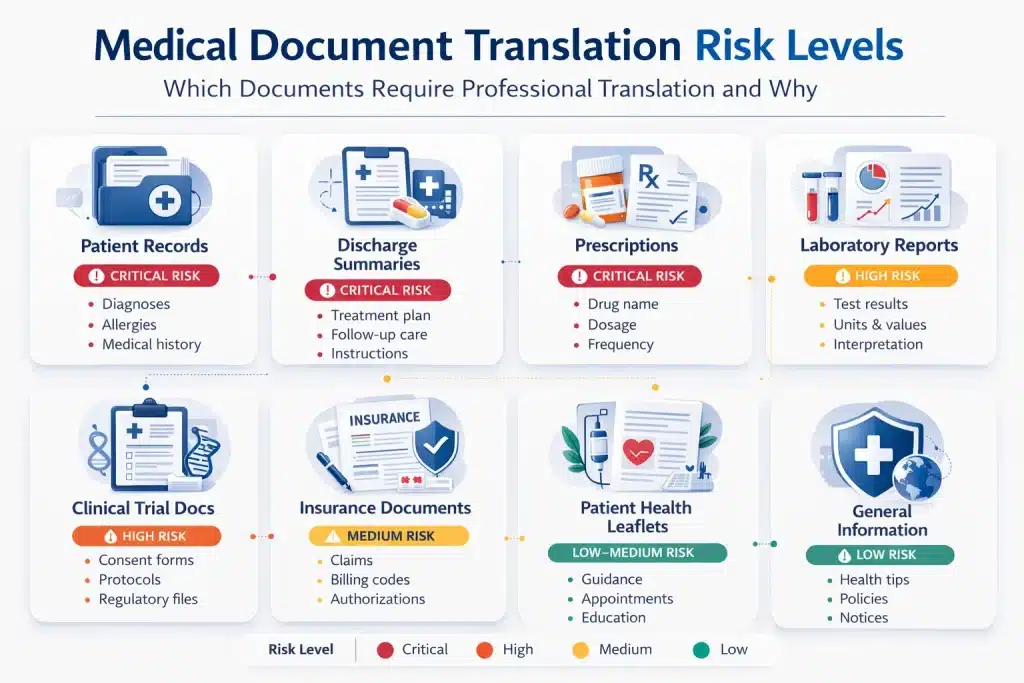
Not all medical documents carry the same translation risk. A mistranslated health leaflet may confuse, but a mistranslated discharge summary or prescription can lead to direct clinical harm. The acceptable translation method depends on the document’s clinical impact, legal status, and regulatory context.
As Federico Federici and Sharon O’Brien (2020) explain in Translation in Cascading Crises, “In healthcare settings, inaccurate translation can have serious consequences for patient safety and quality of care,” highlighting that translation accuracy directly affects clinical outcomes and risk management in medical communication.
| Document type | Examples | Translation risk | Acceptable online or AI use | Certification required |
| Patient records / medical history | Case notes, GP records, allergy lists | Very high. Basis for diagnosis and treatment | No. Professional translation only | Often |
| Discharge summaries | Hospital discharge notes, care instructions | Very high. Continuity of care risk | No. Professional translation only | Often |
| Laboratory results | Blood tests, pathology reports, and biopsy results | High. Clinical interpretation required | No. Professional translation only | Sometimes |
| Medical imaging reports | MRI, CT, X-ray radiology reports | High. Specialist terminology | No. Professional translation only | Rarely |
| Prescription records | Drug names, dosage, administration | Critical. Medication error risk | No. Never use AI tools | Sometimes |
| Informed consent forms | Surgical consent, trial consent | Critical. Legal and regulatory | No. Certified translation only | Always |
| Clinical trial documentation | Protocols, CRFs, investigator brochures | Critical. Regulatory compliance | No. Certified translation only | Always |
| Insurance and billing documentation | EOBs, prior authorisations | Medium to high. Financial and legal | MTPE possible with compliance | Sometimes |
| Pharmaceutical labelling | Drug inserts, safety sheets | High. Patient safety and regulation | No. Professional translation only | Always |
| Medical device documentation | IFUs, technical files | High. Regulatory approval | No. Professional translation only | Always |
| Patient health information | Appointment letters, health leaflets | Medium. Comprehension risk | MTPE possible with review | Rarely |
| Research publications | Journal articles, reviews | Medium. Academic accuracy | MTPE with expert review | No |
Patient Records, Discharge Summaries, and Prescriptions: Why Word-Level Accuracy Is Non-Negotiable
Patient medical records are the most frequently translated healthcare documents and also the most sensitive. These records include diagnoses, allergies, medication histories, and treatment instructions. Even a single translation error can lead to incorrect treatment decisions.
Common failure points in medical record translation include incorrect drug name equivalents, dosage notation errors, mistranslated negation statements such as “no history of,” and misinterpreted medical abbreviations.
Unit conventions also vary between countries. For example, the European notation “1,5 mg” uses a decimal comma, while US notation uses “1.5 mg.” Misinterpreting this difference can lead to a tenfold dosage error.
Prescription records carry the highest clinical risk because four elements must be translated precisely: drug name, dosage, frequency, and administration route. Because of this, prescriptions should never be translated using free online AI tools.
Clinical Trial Documentation: ICH E6(R2), GCP Compliance, and Why Mistranslation Invalidates Studies
Clinical trial translation operates under strict regulatory frameworks, including ICH E6(R2) Good Clinical Practice, the Declaration of Helsinki, and national regulators such as the FDA, EMA, and PMDA. In this context, translation is a documented regulatory process rather than an administrative task.
As the Council for International Organizations of Medical Sciences (CIOMS) and the World Health Organization (2016) state in International Ethical Guidelines for Health-related Research Involving Humans, “Information must be conveyed in language that is understandable to the participant,” emphasizing that informed consent materials must be accurately translated so participants can fully understand the research before agreeing to participate.
Documents requiring translation include protocols, investigator brochures, case report forms, patient questionnaires, safety reports, and informed consent forms. The informed consent form is the most scrutinised document because participants must fully understand the trial before agreeing to participate.
If a consent form translation is inaccurate or too complex for the target population, regulators may determine that valid consent was not obtained. This can delay or invalidate the trial.
To prevent this, clinical trial translation normally includes back translation. The document is translated into the target language and then independently translated back into the source language to verify meaning. This process is documented for regulatory review.
Insurance, Billing, and Cross-Border Healthcare Documentation: The B2B Translation Workflow
Healthcare insurers, hospital networks, and international medical providers handle large volumes of translated documentation as part of their operational workflow. These documents include explanation of benefits statements, prior authorisation requests, referral letters, billing codes, and specialist reports.
Unlike one-off translation requests, these workflows often involve thousands of pages per month and strict turnaround timelines tied to regulatory claim deadlines. Translation services, therefore, operate under volume service agreements rather than per-project pricing.
For these programmes, organisations typically define translation requirements in advance. This includes document volume, language pairs, presence of PHI, required turnaround times, certification needs, and data protection agreements such as NDAs, BAAs, or GDPR data processing agreements.
Why Google Translate, ChatGPT, and Free Online Tools Are Not Safe for Medical Document Translation

Free AI translation tools are designed for general language use. Medical documents require specialist terminology, clinical context awareness, and strict data security controls. Public tools do not provide these safeguards. The risk falls into three categories: translation accuracy, patient data privacy, and regulatory compliance.
| Tool or method | Medical accuracy | Privacy protection | Compliance readiness |
| Google Translate (free) | Moderate to low for specialised terminology | User input may be stored or processed on external servers | Not HIPAA or GDPR compliant |
| ChatGPT or public AI tools | Variable accuracy depending on language and context | User input may be retained for model improvement | No BAA or healthcare compliance framework |
| DeepL free version | Higher general accuracy but limited medical terminology support | Data processing on external infrastructure | No healthcare compliance guarantees |
| Professional medical translation | Specialist medical translators and QA review | Secure encrypted workflows | HIPAA and GDPR compliant with contracts |
Accuracy Failure: What Clinical Studies Say About AI Tool Error Rates in Medical Translation
Clinical research shows that free AI tools produce significant error rates in medical translation. A 2024 Oregon Health and Science University study comparing ChatGPT 3.5 and Google Translate for paediatric urology instructions found sentence-level error rates ranging from 3.8 percent to over 40 percent depending on language.
ChatGPT produced 3.8 percent errors in Spanish but over 35 percent in Russian. Google Translate reached 41.6 percent error rates in the same language pair. Another 2024 review of Haitian Creole discharge instructions identified clinically significant translation errors from both systems that professional translators did not produce.
Medical language requires specialised knowledge of dosage conventions, abbreviations, and clinical context. Even a small error rate becomes unacceptable when translating medication instructions, diagnoses, or allergy information.
HIPAA Violations: Why Google Translate and ChatGPT Cannot Legally Handle PHI
HIPAA requires any organisation handling Protected Health Information on behalf of a healthcare provider to sign a Business Associate Agreement. This contract defines how patient data is protected, stored, and accessed.
Free AI translation tools do not provide this legal framework. Google Translate does not sign BAAs for its public service, and standard ChatGPT access does not include a HIPAA-compliant agreement. As a result, uploading patient identifiable information to these platforms violates HIPAA requirements.
The compliance gaps include a lack of a Business Associate Agreement, potential storage of submitted text on external servers, the absence of access controls, and no audit logs tracking who accessed patient data.
HIPAA penalties range from 100 dollars to 50,000 dollars per violation, with annual caps reaching approximately 1.9 million dollars per violation category.
GDPR and Patient Data: What European Healthcare Organisations Must Require from Any Translation Vendor
Under GDPR, healthcare data is classified as a special category of personal data requiring strict protection. Any translation vendor processing medical records must operate under a legally binding Data Processing Agreement before receiving patient information.
A compliant vendor must define the purpose of processing, data security measures, permitted sub-processors, and breach notification procedures. Organisations must also follow the data minimisation principle by removing unnecessary patient identifiers when possible.
Public AI tools typically process content on global cloud infrastructure and do not provide formal GDPR processing agreements for free access. Transmitting patient records through these systems can therefore create a GDPR Article 9 compliance risk.
Before sending any medical document for translation, healthcare organisations should confirm that the vendor signs a GDPR compliant DPA and specifies where patient data is processed.
Terminology Gaps: Why Medical Abbreviations, Brand Names, and Regional Variants Break AI Tools
Medical translation challenges arise from terminology systems that vary by country, specialty, and regulatory framework. AI translation models often struggle with these variations.
Common failure areas include medical abbreviations such as BID, PRN, NPO, and STAT, which do not always have direct equivalents in other languages. Drug naming conventions also vary between international nonproprietary names and regional brand names.
Anatomical terminology can appear similar across languages because of Latin roots, but patient-facing terms differ by region. Measurement conventions add further complexity because medical values may require conversion between metric and imperial systems.
Professional medical translation workflows mitigate these risks by applying client-specific glossaries and translation memories built from previously validated translations.
What Safe Online Medical Document Translation Actually Looks Like: The Compliant Workflow
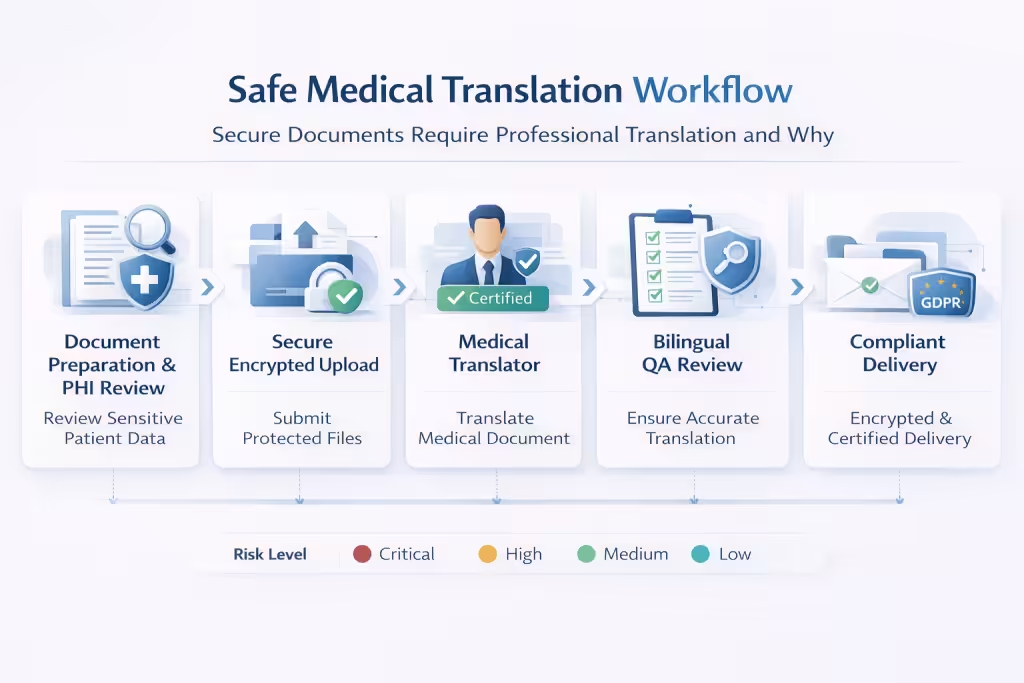
Online medical document translation can be performed safely when the workflow includes proper compliance, security, and quality controls. The key question is not whether translation occurs online, but whether the workflow meets healthcare data protection standards and clinical accuracy requirements.
| Workflow stage | Safe professional workflow | Unsafe approach |
| Document submission | Secure encrypted upload via compliant portal | Uploading documents to public AI tools |
| Data protection | BAA and GDPR Data Processing Agreement in place | No legal data protection agreement |
| Translation process | Qualified medical translators with QA review | Raw machine translation without review |
| File delivery | Encrypted file transfer and controlled retention | Files sent via unsecured email or stored indefinitely |
Step 1–2: Document Preparation, PHI Handling, and the Pre-Submission Compliance Checklist
Step 1 begins with reviewing the document for Protected Health Information. Patient identifiers such as names, dates of birth, addresses, medical record numbers, diagnoses, and treatment details must be identified before submission.
If the translation does not require patient identity information, these elements should be redacted. This aligns with GDPR’s data minimisation principle and reduces privacy risk.
Step 2 involves verifying the translation vendor’s compliance framework. Healthcare organisations should confirm that the vendor signs Business Associate Agreements for HIPAA compliance and GDPR Data Processing Agreements for EU patient data.
Secure file transfer methods, documented retention policies, translator confidentiality agreements, and information security standards such as ISO 27001 and ISO 17100 should also be verified before submitting any document.
Step 3–4: Medical Translator Qualification, Terminology Glossary, and QA Review Standards
Professional medical translation requires translators with both linguistic and domain expertise. Qualified translators should demonstrate native proficiency in the target language, specialised knowledge in the relevant medical discipline, familiarity with the healthcare system of the target market, and professional certification or membership such as ATA, ITI, or CCHI.
Domain matching is critical. Clinical records require translators familiar with clinical practice, while pharmaceutical documents require pharmacology expertise. Medical device documentation often requires translators with biomedical engineering knowledge.
Quality assurance standards include independent bilingual review, subject matter expert validation where required, and back translation for clinical trial documentation.
A terminology glossary and translation memory ensure consistent terminology across all documents. For regulated submissions, the entire QA process must be documented and auditable.
When MT Post Editing Is Acceptable for Medical Content and the Conditions Required
Machine Translation Post-Editing can be used in limited medical translation scenarios when strict conditions are met. The content must not contain identifiable patient data unless processed within a secure enterprise translation environment covered by a BAA or DPA.
The machine translation output must also be reviewed by a qualified medical translator rather than a general linguist.
MTPE is appropriate for lower-risk content such as general health education materials, internal healthcare communications, administrative notices, and policy documentation. It can reduce translation costs by roughly 35 to 55 percent compared with full human translation.
However, MTPE is not suitable for prescriptions, discharge summaries, diagnoses, consent forms, or any regulatory submissions. These documents require fully human translation with formal quality assurance procedures.
The 5 Biggest Accuracy Challenges in Medical Document Translation and How Professionals Handle Them
Medical translation errors are rarely random. They tend to occur around predictable linguistic and technical failure points such as terminology interpretation, cultural context, regulatory language, and data formatting.
Understanding these risk areas helps healthcare organisations evaluate translation vendors and implement quality controls that prevent clinical or regulatory mistakes.
Complex Medical Terminology: Standardisation, False Cognates, and Language-Specific Clinical Conventions
Medical terminology often appears consistent across languages because many terms share Latin or Greek roots. In practice, this similarity can create misleading translations. False cognates and partially equivalent terms may look correct while conveying a different clinical meaning.
Examples include words such as “intoxication,” which in English typically refers to poisoning but may indicate general substance exposure in other European languages. Anatomical terminology can also vary between clinical and patient-facing registers.
Eponym-based terms such as Parkinson’s disease or Crohn’s disease may either remain unchanged or be replaced by descriptive equivalents depending on the language.
Professional medical translation workflows address these risks through custom terminology glossaries developed from validated translations and reviewed by subject matter experts in the target healthcare system.
Cultural Sensitivity and Healthcare Context: When Literal Translation Creates a Communication Failure
A technically correct translation can still fail if it ignores the cultural context in which healthcare communication occurs. Concepts of illness, patient autonomy, and family involvement in medical decision making differ significantly between cultures.
For example, many healthcare systems assume individual patient decision-making, while other cultures rely on family involvement when approving treatment or clinical trial participation. Patient-reported outcomes such as pain scales or quality of life questionnaires may also require cultural adaptation to ensure patients interpret questions consistently.
In clinical trials, this process is known as linguistic validation and follows international guidelines such as those issued by ISPOR. It involves translation, cultural adaptation, and validation testing rather than simple direct translation.
Legal and Regulatory Language in Medical Documents: Precision Requirements Beyond Clinical Accuracy
Some medical documents function simultaneously as clinical records and legal instruments. Examples include informed consent forms, litigation medical reports, insurance claim documentation, and regulatory submission materials.
In these cases, the translation must preserve both clinical meaning and legal validity. Consent language that is legally acceptable in one jurisdiction may require structural changes to remain legally effective in another. Regulatory agencies such as the FDA and EMA also use specific terminology conventions that must be reflected in translated submissions.
For litigation or immigration documentation, the wording of clinical certainty statements can have legal consequences. Professional translation workflows, therefore, involve translators experienced in both medical and legal terminology or legal review in the target jurisdiction.
Formatting and Layout Integrity: Why Tables, Units, and Numerical Data Create Translation Errors
Medical documents contain large amounts of structured information, including tables of laboratory values, dosing schedules, and diagnostic measurements. Translation errors in these elements often occur during formatting rather than language translation.
Table structure can be misinterpreted if cells are translated individually without considering the relational layout of the data. Numerical precision must also be preserved because small differences in decimals can change clinical interpretation.
Unit conventions vary between healthcare systems. For example, blood glucose values may be recorded in millimoles per litre in Europe but milligrams per decilitre in the United States. Date formatting differences between regions can also create confusion in medical records.
Professional medical translation workflows include format-aware editing that preserves table structure, verifies unit conventions, and validates all numerical data against the source document.
Decision Framework: Which Medical Translation Method Is Right for Your Use Case?
The correct translation approach for a medical document depends on four variables: clinical risk, presence of patient identifiable information, regulatory requirements, and translation volume. The framework below helps healthcare organisations select the appropriate method for common B2B translation scenarios.
| Use case | PHI present | Clinical risk | Regulatory requirement | Acceptable method | Certification required |
| Patient records for cross-border care | Yes | Very high | HIPAA or GDPR | Certified human TEP with BAA or DPA | Often |
| Clinical trial informed consent form | Yes | Critical | GCP, ICH, EMA, or FDA | Certified human translation with back translation | Always |
| Prescription records | Yes | Critical | HIPAA or GDPR | Certified human translation only | Often |
| Discharge summary for inbound patient | Yes | Very high | HIPAA or GDPR | Professional human TEP with BAA or DPA | Sometimes |
| Laboratory results for specialist consultation | Yes | High | HIPAA or GDPR | Professional human TEP with BAA or DPA | Rarely |
| Insurance or EOB documents with PHI | Yes | Medium to high | HIPAA or GDPR | MTPE in secure compliant environment | Sometimes |
| Pharmaceutical package insert | No | High | EMA or FDA regulation | Certified human TEP | Always |
| General patient health information | No | Low to medium | WCAG or ADA accessibility | MTPE with human review | No |
| HR health policy documents | No | Low | None | MTPE acceptable | No |
| Medical research publication for internal review | No | Medium | None | MTPE with subject matter expert review | No |
When Certified Medical Translation Is Required vs When It Is Strongly Recommended
Certified translation refers to a formal statement from the translator or translation provider confirming that a translation is complete and accurate. In medical contexts, this certification is often required for documents submitted to authorities or used in legal proceedings.
Notarised translation adds a notary verification of the translator’s identity and signature, while sworn translation applies in civil law countries where officially authorised translators produce legally valid documents.
Certified medical translation is typically required for immigration medical records, clinical trial documentation submitted to regulators, cross-border insurance claims, medical evidence used in litigation, and health certificates for international travel or employment.
For clinical care between healthcare providers, certification may not always be legally required, but professional human translation with quality assurance remains strongly recommended.
Medical Translation Cost Benchmarks: Per Word, Per Page, and Volume SLA Pricing in 2026
Medical translation costs are typically higher than general translation because they require specialist domain expertise and stricter quality assurance processes.
Pricing varies depending on document complexity, regulatory requirements, and whether certification or back translation is required.
| Document type or service | Pricing model | Typical range | Notes |
| Patient records and discharge summaries | Per word | €0.20 to €0.35 | Standard major language pairs with human TEP |
| Clinical trial documentation | Per word | €0.25 to €0.45 | Includes back translation and regulatory QA |
| Pharmaceutical label or package insert | Per word | €0.22 to €0.40 | Regulatory terminology expertise required |
| Medical device documentation | Per word | €0.18 to €0.30 | Technical and medical domain overlap |
| MTPE medical content | Per word | €0.10 to €0.18 | Requires qualified medical post editor |
| Certified medical translation | Per page | €55 to €150 | Includes certification statement |
| Notarisation add on | Per document | €20 to €75 | Notary authentication fee |
| Back translation for trials | Per word | €0.12 to €0.20 additional | Separate translator and comparison report |
| Rush delivery surcharge | Percentage | +25 to +60 percent | Standard turnaround is 5 to 10 business days |
For institutional buyers such as hospitals, insurers, CROs, and pharmaceutical companies translating more than 10,000 words per month, service level agreements usually reduce per-word pricing by 15 to 25 percent.
These agreements typically define compliance requirements such as BAA or GDPR DPA coverage, QA tiers, turnaround times, and translation memory ownership.
Translate Medical Documents Online Safely — Compliant, Accurate, and Professionally Certified
Circle Translations provides HIPAA and GDPR compliant medical document translation in more than 120 languages. All translations are completed by qualified medical translators with relevant clinical or regulatory expertise, not by general linguists reviewing raw machine output.
Every medical translation engagement includes:
✓ HIPAA Business Associate Agreement signed before any PHI is submitted
✓ GDPR compliant Data Processing Agreement for EU patient data
✓ Medical translator matched to the document domain, such as clinical, pharmaceutical, regulatory, or insurance
✓ Custom medical terminology glossary applied from the first project
✓ Independent second linguist QA review as standard, with back translation available for clinical trial submissions
✓ Encrypted secure file transfer for all documents
✓ Confidentiality agreements signed by all translators, editors, and project managers
✓ Certification statements provided for documents requiring official submission
Submit your medical document for a confidential, itemised quote.
Get a Compliant Medical Translation Quote → | View Medical Translation Services →
Frequently Asked Questions – Translate Medical Documents Online
Can ChatGPT translate medical records safely and accurately?
No. ChatGPT should not be used to translate medical records for clinical or official purposes. It can produce medical terminology errors and does not provide HIPAA or GDPR compliant data protection for patient information.
Is Google Translate HIPAA compliant for medical documents?
No. Google Translate is not HIPAA compliant. The service does not provide a Business Associate Agreement, which is required when processing Protected Health Information.
What is a Business Associate Agreement (BAA), and why does medical translation require one?
A BAA is a legal contract required under HIPAA between a healthcare organisation and any vendor handling Protected Health Information. Medical translation providers must sign a BAA before processing patient records.
How do I know if a medical translation is accurate enough to use clinically?
Clinically reliable translations are completed by qualified medical translators and reviewed by a second linguist. For high-risk documents, additional QA, such as back translation, may be used.
Can I redact patient information before sending a medical document for translation?
Yes. Patient identifiers such as names or ID numbers can be replaced with placeholders if they are not required for translation accuracy. This reduces privacy risk under GDPR and HIPAA.
How long does professional medical document translation take?
Most medical documents, such as patient records or discharge summaries, take two to five business days. Certified translations may take three to seven days, depending on length.
Does Circle Translations provide certified medical translation for visa and immigration purposes?
Yes. Circle Translations provides certified medical translations accepted by immigration authorities such as USCIS and UKVI.
What is the difference between medical translation and medical interpretation?
Medical translation covers written documents. Medical interpretation refers to real-time spoken communication between healthcare providers and patients.
What happens if a medical translation contains an error, and who is liable?
With a professional translation provider, liability is defined in the service contract and supported by professional indemnity insurance. Free AI tools provide no liability protection.